|
12/31/2023 0 Comments Transition metals reactivity
Looking for something specific in this course? The Resource Index compiles links to most course resources in a single page. Lecture 29: Transition Metals: Crystal Field Theory Part II For example, pure iron will react with water, and oxygen in the air, and will rust, but this process is slow compared to the reaction of some other elements. For example, the HgC bond of (MeHg) + cation is indefinitely stable in aqueous H 2 SO 4 solution in air. On the contrary the late transition metal organometallic compounds are much less reactive and stable. Lecture 28: Transition Metals: Crystal Field Theory Part I Similar high reactivity was also observed in case of the early transition metal organometallic compounds particularly of Ti and Zr. Lecture 27: Introduction to Transition Metals Lecture 26: Chemical and Biological Oxidations 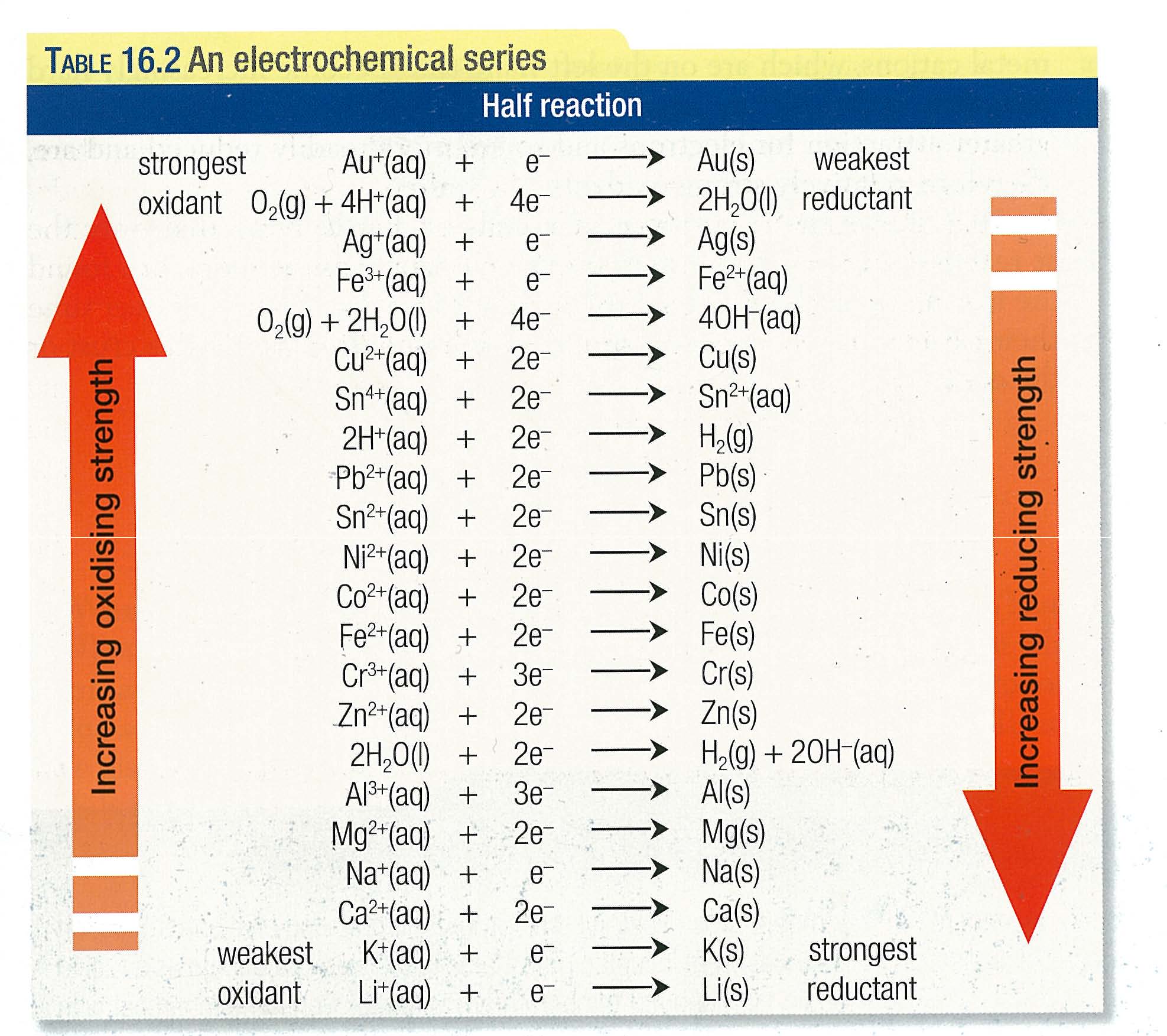
Lecture 25: Oxidation-Reduction and Electrochemical Cells By the end of the unit, viewers should be able to calculate electrochemical cell potentials, identity which compound is the better oxidizing agent, draw energy diagrams that explain why one cobalt compound is blue and another red, and explain how a chelator works to treat acute lead poisoning. Viewers can observe some of these special properties for themselves as spectacular color changes afforded by solutions of nickel and cobalt salts are demonstrated. Revisiting the concept of atomic orbitals, we discuss how the occupation of electrons in d-orbitals gives rise to the special properties of d-block metal-containing complexes. Whereas metals in group I and II of the periodic table are only oxidized by one and two electrons, respectively, transition (d-block) metals are capable of achieving multiple oxidation states. In Unit IV, we continue to consider chemical reactions but now focus on reactions that involve the loss (oxidation) and gain (reduction) of electrons. But along the period, the reactivity of the elements is not dependent on the oxidation state.Unit IV: Transition Metals & Oxidation-Reduction Reactions Note: The lanthanide contraction is seen in the 5d-series of the d-block elements, so the ionisation enthalpy as well as the reactivity of the elements of 3d is not affected by it.Īlso, the oxidation state of the 3d-series elements corresponds to the loss of electrons till Mn, but after that again the number of oxidation states decreases, with the decrease in the atomic size. Therefore, the reactivity of the transition elements decreases almost regularly from Sc to Cu because of option (B)- regular increase in ionisation enthalpy. As along the period, with the decrease in the atomic size, the electronegativity increases, so the enthalpy of hydration of the cation decreases, making the element unreactive. This increase in the ionisation enthalpy thus, leads to the decrease in the reactivity of the element. Thus, along the period the ionisation enthalpy increases. As more the nuclear charge, more the attraction of the electrons, and higher is the ionisation energy needed to form the ion. It is related to the force of attraction between the nucleus and the electrons. 
Then, the ionisation energy is the amount of energy required to remove the outermost electron from the orbit in the isolated gaseous state of the atom to form the ion. The elements from Scandium to Copper belong to the First transition series of the d-block, where with the increase in the atomic number, the nuclear charge increases, and the atomic radius decreases along the period. The energy to remove an electron required increases, which in turn determines the reactivity of the element. Hint: As along the period, the size decreases along with the increase in the electronegativity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
